![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


![H H+ + H H H H Proton donor (acid) Proton acceptor (base) Hydroxide ammonia water ion H]. + mai H H H](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1702/0/2/2/2336572cc599721a1702022231301.jpg)
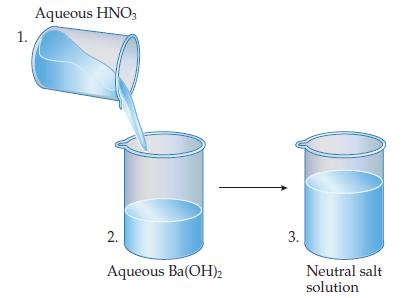
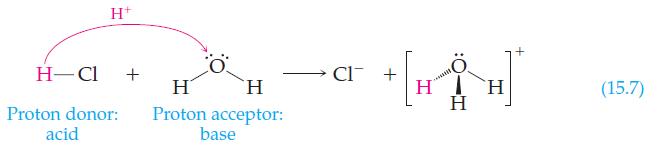
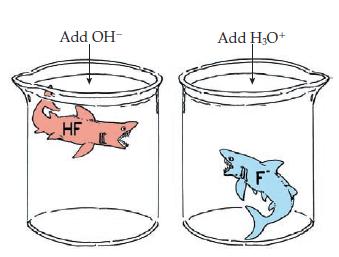
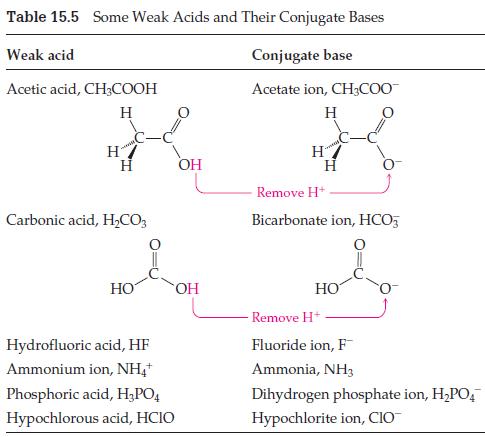
![Table 15.4 The pH Scale [HO+] [OH-] 10 10-14 10-1 10-13 10- 10-3 10-4 10-5 10-6 10-7 10-8 10-9 10-10 10-11](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/9/4/9/2706571af56d7b581701949270028.jpg)