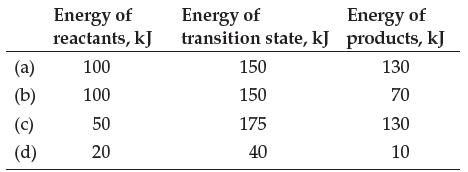
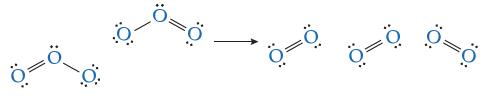
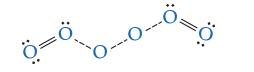
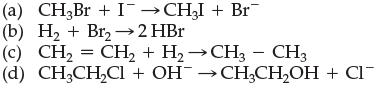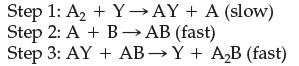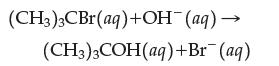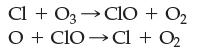![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()



![H,O2(aq)+3I (aq)+2H*(aq)I3(aq)+2H,O(1) Experiment [HO] [1] 1 0.010 M 0.010 M 2 0.020 M 0,010 M 3 0.010 M](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/8/5/8/96765704e971cb161701858967100.jpg)
![Experiment 1 2 3 [A] 0.0125 M 0.0250 M 0.0125 M [B] 0.0253 M 0.0253 M 0.0506 M Rate of disappearance of A](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/8/5/9/03565704edb3c9401701859034682.jpg)
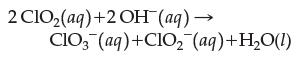
![Experiment 1 23 2 [C10] 0.060 M 0.020 M 0.020 M [OH-] 0.030 M 0.030 M 0.090 M Rate (M/s) 0.024 84 0.002 76](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/8/5/9/10265704f1e898191701859100897.jpg)
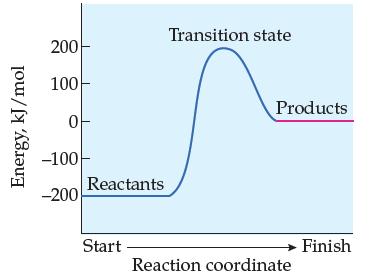
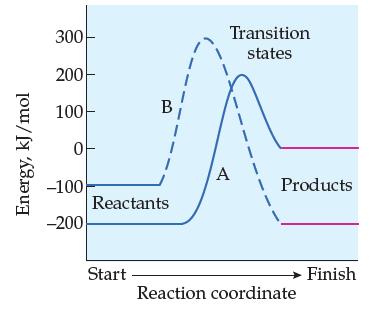
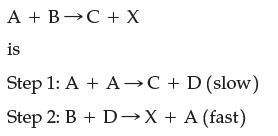
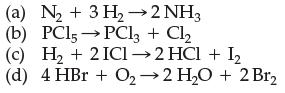
![A + A A is Rate = K[A][BC] Two mechanisms have been proposed for the reaction: Step 1: A + BAB (slow) Step](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/8/6/0/500657054943bb4c1701860498083.jpg)