![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


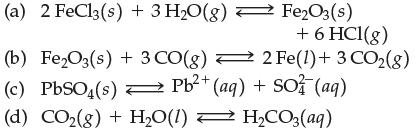
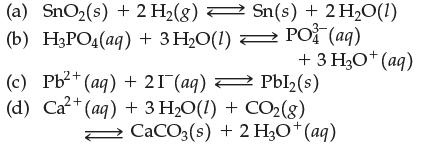
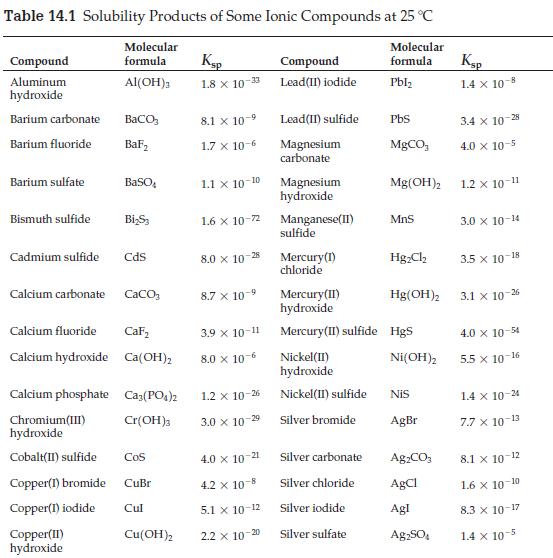

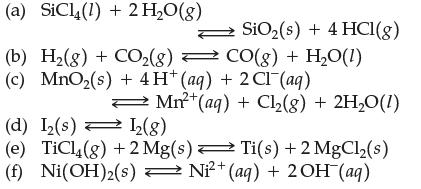
![[A]x [B] [C]X [D] Keq (a) A2+ B3 (b) 2A + 3B (c) 3C + 2D (d) A+B (e) 2C + 3D C3 + D 3C + 2D 2A + 3B C + D 3A](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/9/3/2/35465716d4229b521701932351076.jpg)
![Concentration [NO] [NO] 0 1 2 3 4 5 6 7 8 9 10 11 12 13 Minutes](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/9/3/3/3736571713d4eb6b1701933373265.jpg)
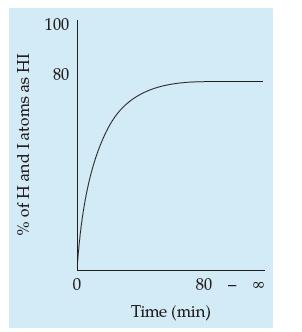
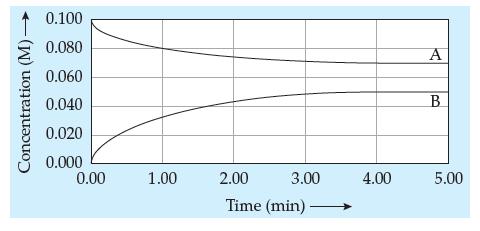
![| Ai H+ H + ]:OH H HI (15.4)](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1702/0/2/0/4976572c591e1ef81702020496615.jpg)
![Solution 1 2 234 4 5 67 7 8 9 10 1122 13. 14 15 [H3O+] 10 (same as 1.0 10 M = 1.0 M) 10-1 10- 10-3 104 10-5](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1702/0/2/2/3196572ccaf342ef1702022316109.jpg)
![(a) [HO+] = 2 M [OH-] = ? (b) [HO+] = ? [OH-] - 4 M =](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1702/0/2/2/3606572ccd8cb2d51702022358434.jpg)