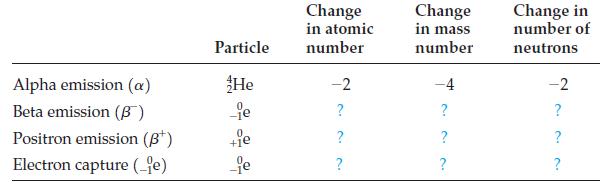
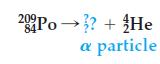![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


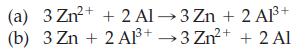
![Experiment 1 1234 2 4 Initial concentration (mol/L) [NO] 0.0060 0.0060 0.0010 0.0020 [H] 0.0010 0.0020 0.0060](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/8/6/2/85765705dc91f16c1701862855380.jpg)
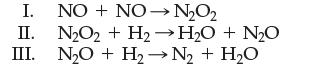
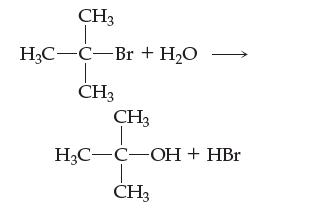
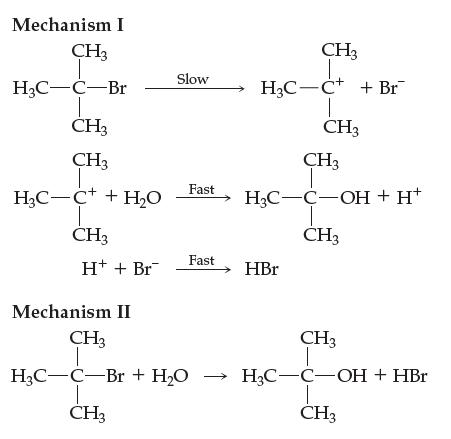
![[NO] = 1.00 x [N] = x [0] = x](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/9/4/0/73065718dfad3b5d1701940731315.jpg)
![Keq [N][0] [N0] [x][x] [1.00 x] 0.456](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/9/4/0/75165718e0fe3afc1701940752430.jpg)