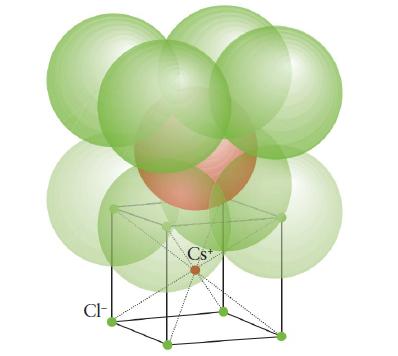
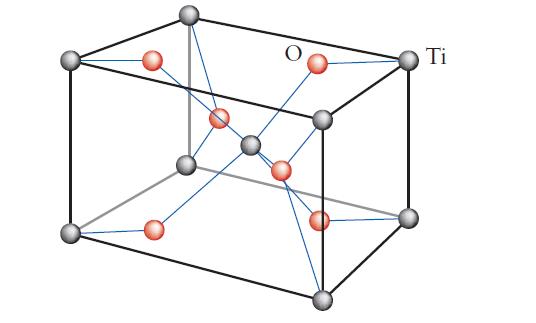
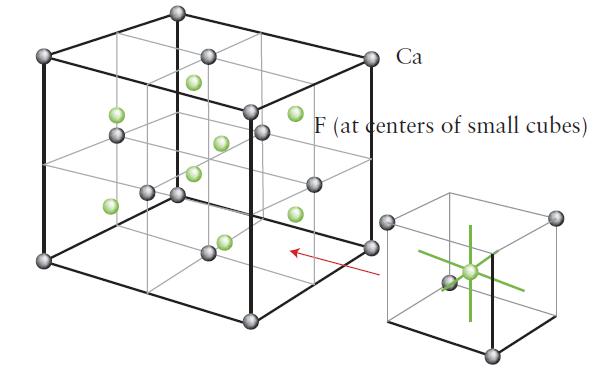
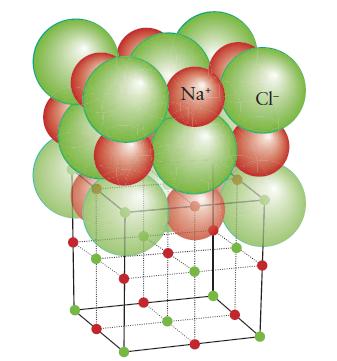
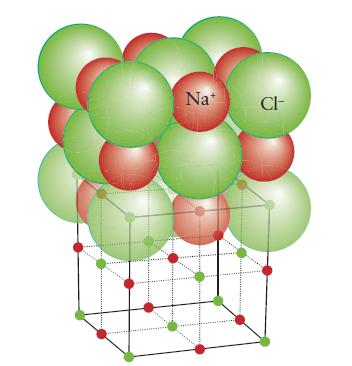
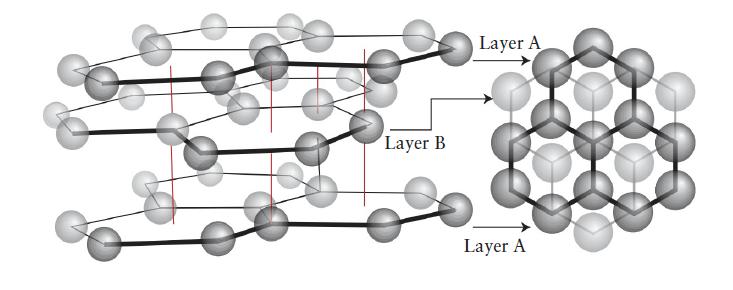
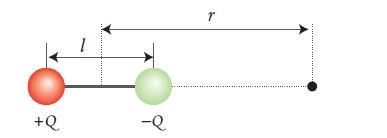
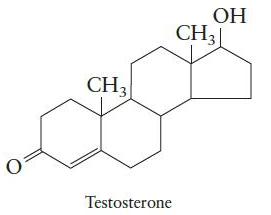
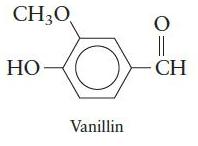
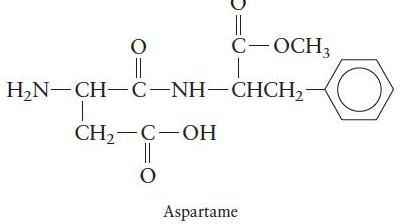
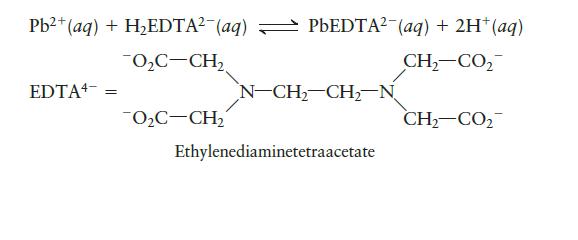
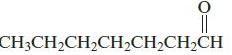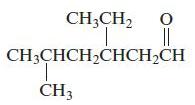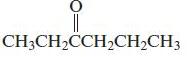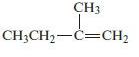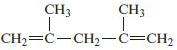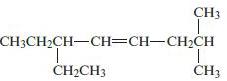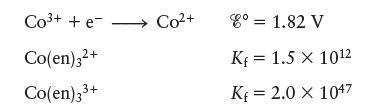Chemical Principles 8th Edition Steven S. Zumdahl, Donald J. DeCoste - Solutions
Unlock the potential of your chemistry studies with the "Chemical Principles 8th Edition" by Steven S. Zumdahl and Donald J. DeCoste. Dive into a comprehensive collection of questions and answers, complete with an online solutions manual and detailed answers key. Whether you're tackling solved problems or seeking chapter solutions, our resource offers step-by-step answers ideal for mastering the textbook content. Access the instructor manual and test bank for in-depth understanding and practice. Enjoy the convenience of a solutions PDF and explore the textbook's insights with free download options, enhancing your learning journey effortlessly.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()