![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


![Energy of Quanta, kJ mol- 0 203 308 349 362 Electron Configuration [Ne]3s [Ne]3p [Ne]4s [Ne13d [Ne]4p](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/8/5/0/2406551a8009ea611699850238049.jpg)

![[Ar] NNNNN 3d 4s 4p](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/6/8/0/760654f11f806478Screenshot_2023-11-11_103152.png)
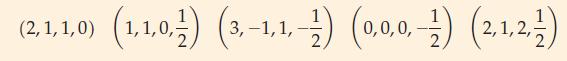
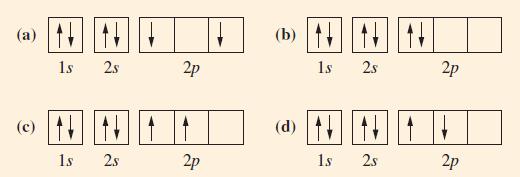
![[Ar] NNNNNNN 3d 4s 4p](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/6/7/6/314654f009ab41de1699676314570.jpg)