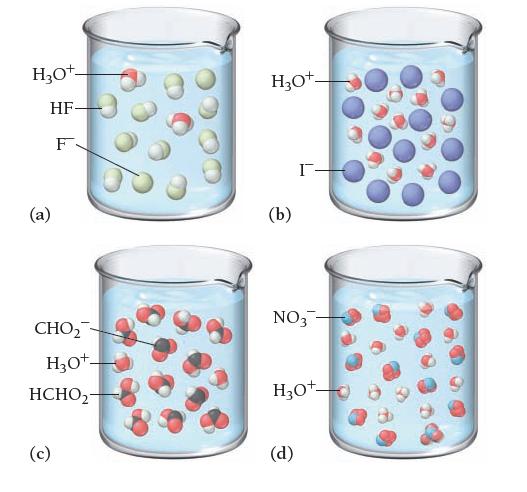
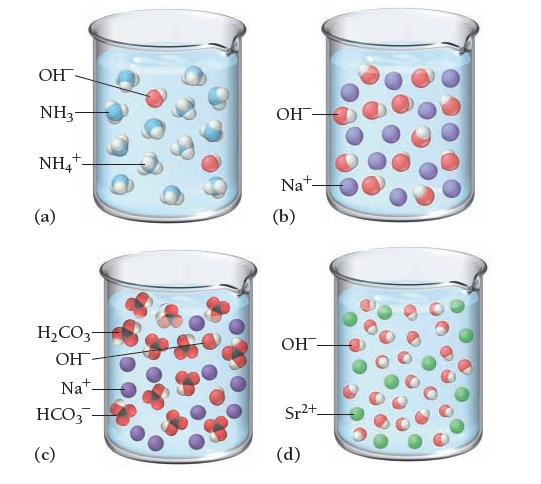
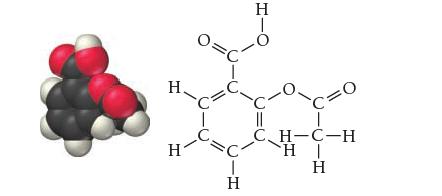
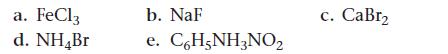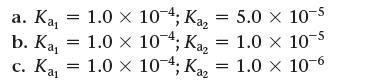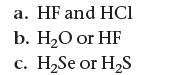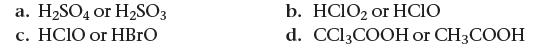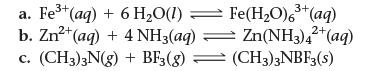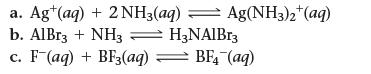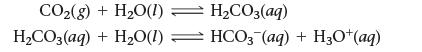![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


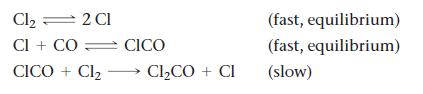
![a. [H3O+] 1.2 x 10-8 M C. [H3O+] = 3.5 x 10- M = b. [H3O+] = 8.5 x 10-5 M](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/2/9/0/6206558603c884e01700290619671.jpg)
![a. [H30] = 1.7 x 10-8 M C. [H3Ot] = 2.2 x 10-6 M b. [H3O] = 1.0 107 M](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/2/9/0/6686558606ce70be1700290661395.jpg)
![[H3O+] 3.7 X 10- [OH-] 1.6 X 10-11 pH 3.15 11.1 Acidic or Basic](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/2/9/0/735655860af652e11700290734231.jpg)
![[H3O+] 3.5 x 10-3 1.8 x 10 [OH-] 3.8 x 10-7 pH 7.15 Acidic or Basic](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/2/9/0/759655860c76c45f1700290758410.jpg)
![[H3O] = 0.044 M [H3O] = 0.045 M [H3O] = 0.046 M](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/2/9/0/83365586111e291b1700290833089.jpg)