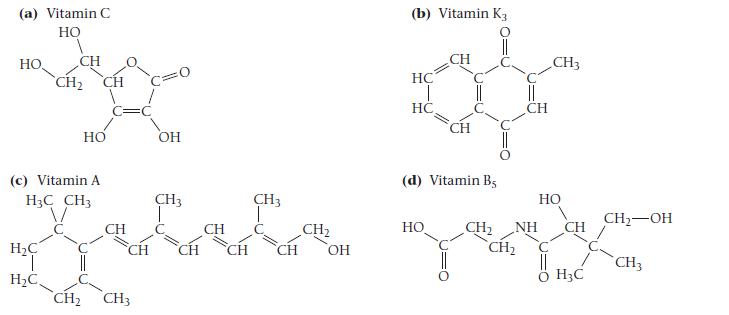
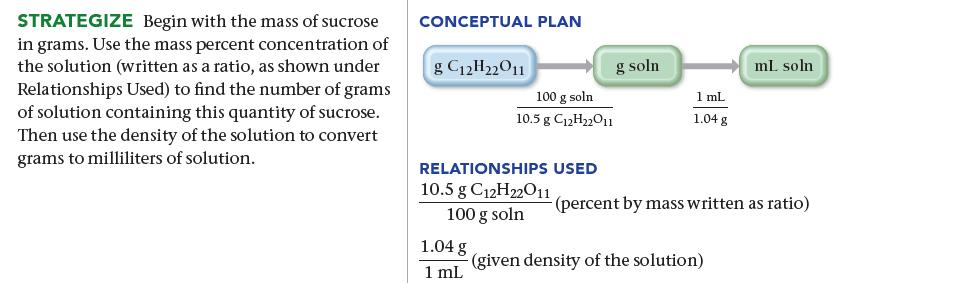
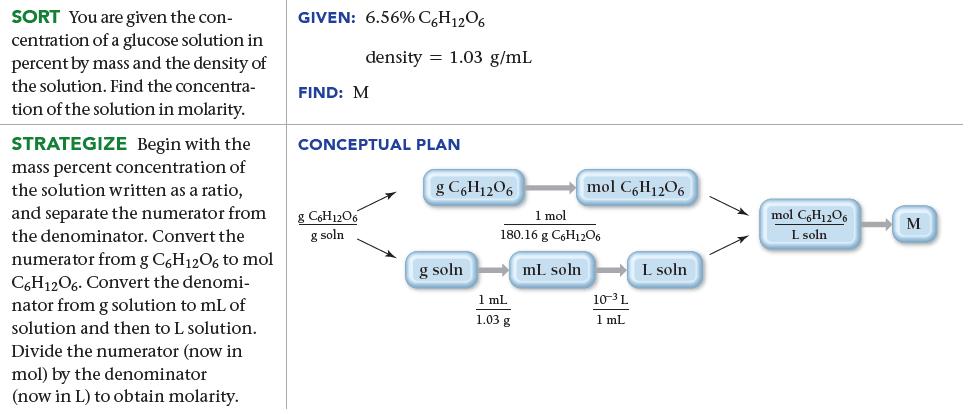
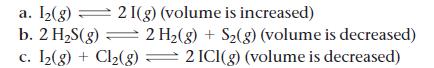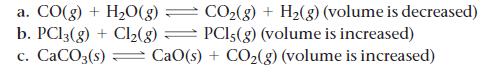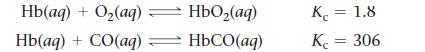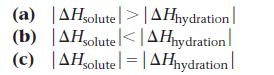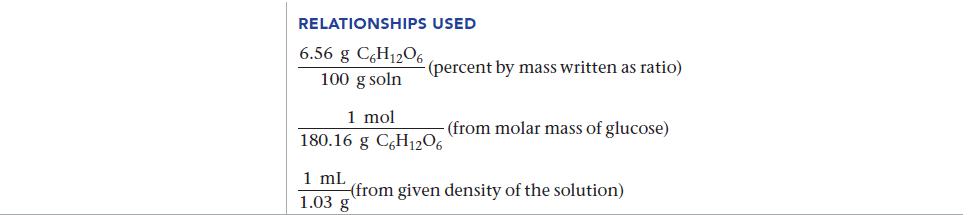![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


![PC15 (8) PC13 (1) + Cl(g) Ke [PC13][C1] [PC]s]](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/2/1/4/67865573796eacf31700214678519.jpg)