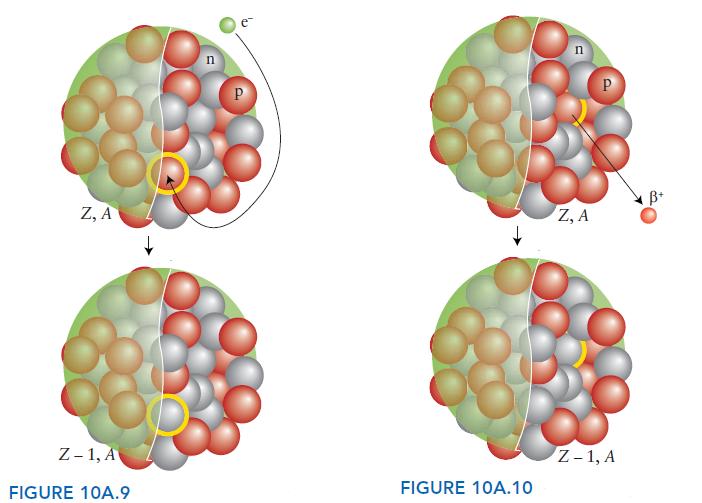
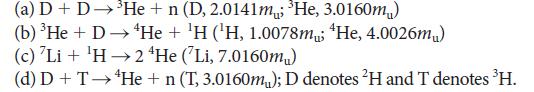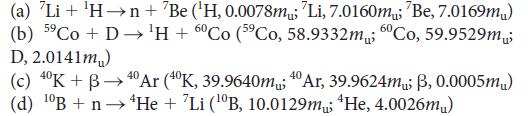Chemical Principles The Quest For Insight 7th Edition Peter Atkins, Loretta Jones, Leroy Laverman - Solutions
Discover in-depth explanations and comprehensive solutions for the "Chemical Principles: The Quest For Insight, 7th Edition" by Peter Atkins, Loretta Jones, and Leroy Laverman. Our online platform offers the complete answers key and solutions manual, including solved problems and questions and answers for each chapter. Access the test bank and chapter solutions with step-by-step answers that align with the instructor manual. This textbook’s solutions are available in a convenient solutions PDF format, ensuring you can study efficiently. Additionally, explore free download options for these resources, enhancing your learning experience.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


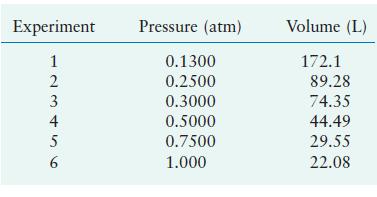
![[NH3] 3.1 x 10- mol/L [N] = 8.5 x 10 mol/L [H] = 3.1 x 10- mol/L](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1705/9/2/7/58465ae63a06c1501705927585383.jpg)
![-3 a. [NH3]0= 1.0 10-3 M; [Nlo = 1.0 x 105 M; [Hlo = 2.0 10- M b. [NH3]0 = 2.00 x 10-4 M; [Nl0 = 1.50 x 105](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1705/9/2/7/76465ae645466a191705927764895.jpg)