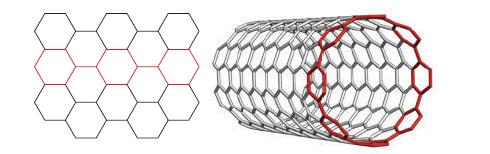
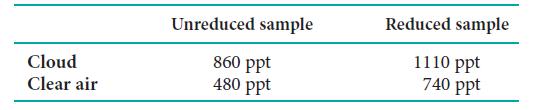
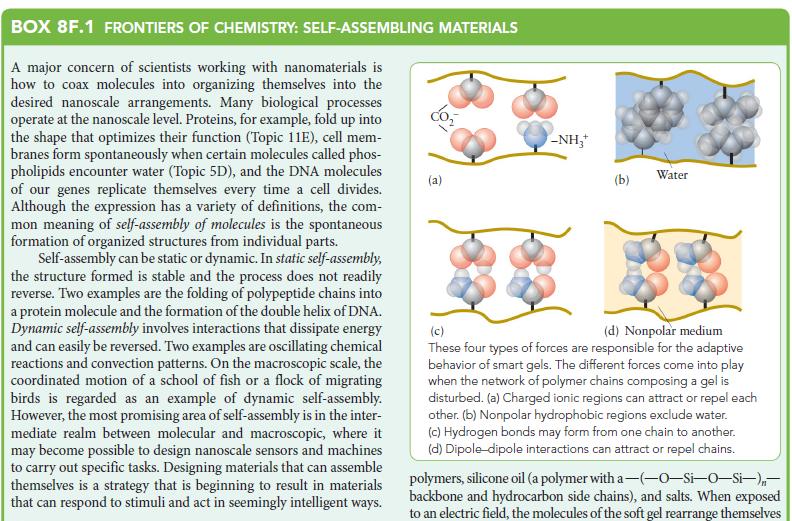
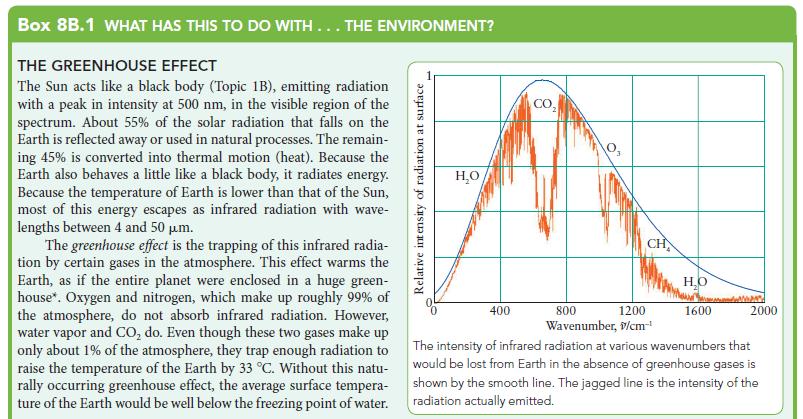
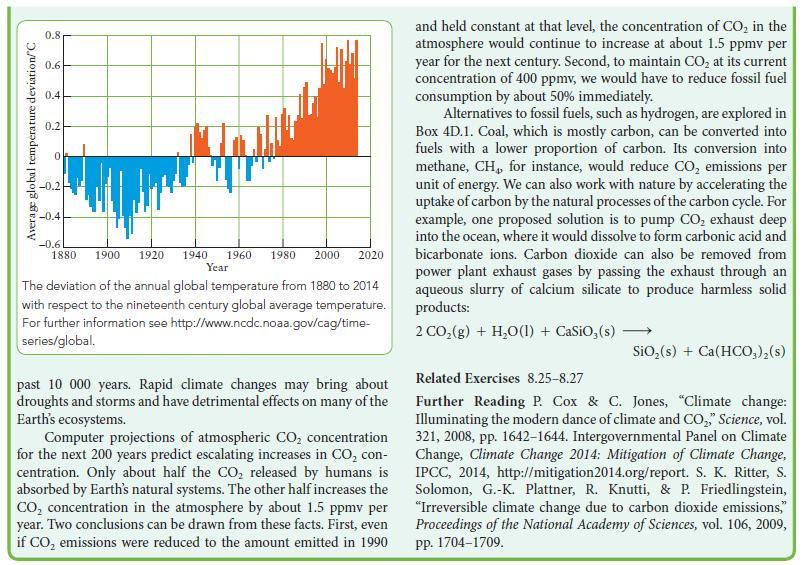
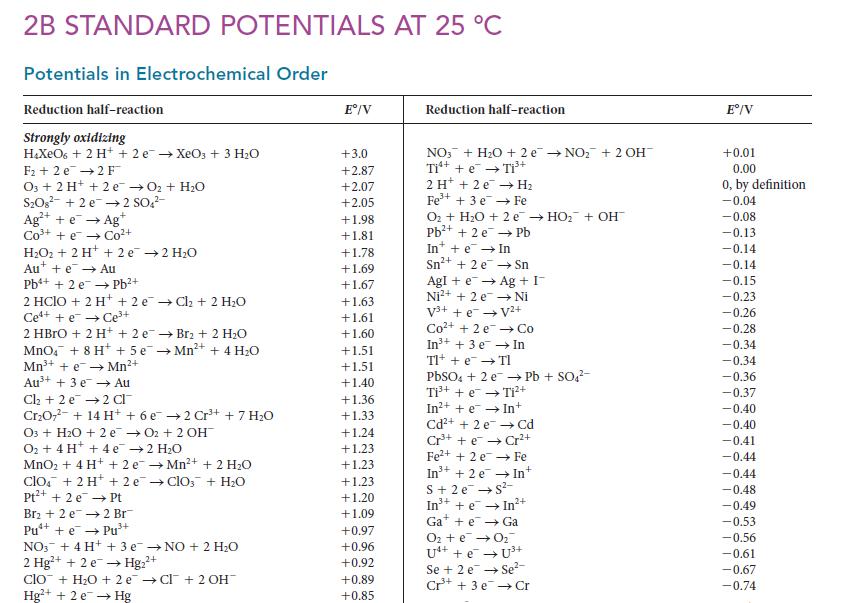
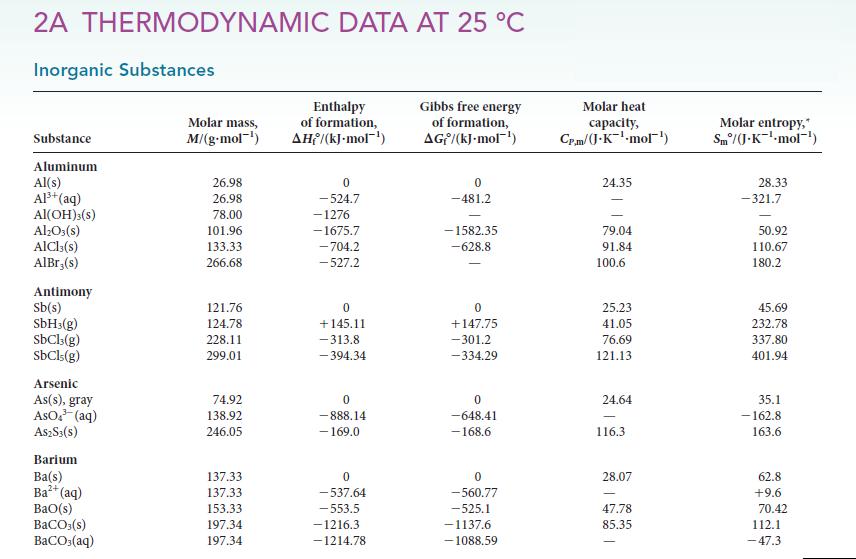
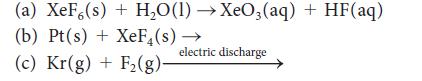![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


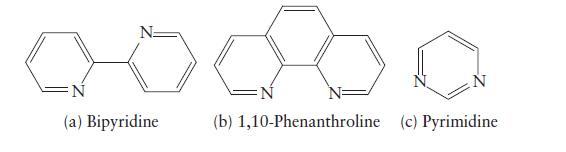
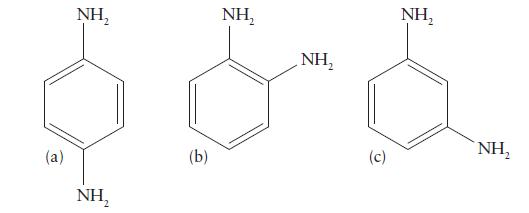
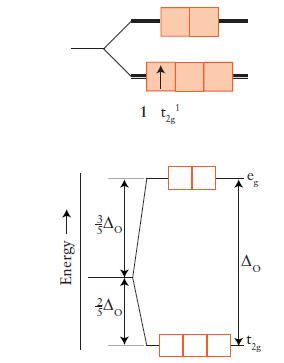
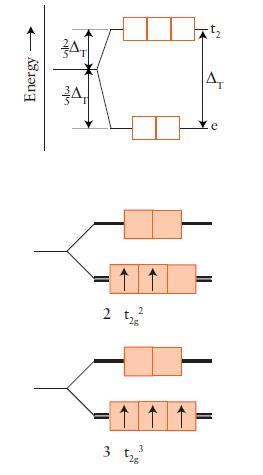
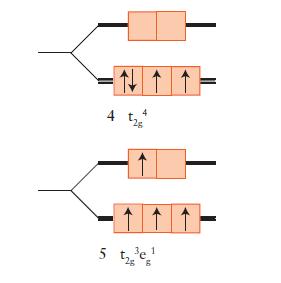
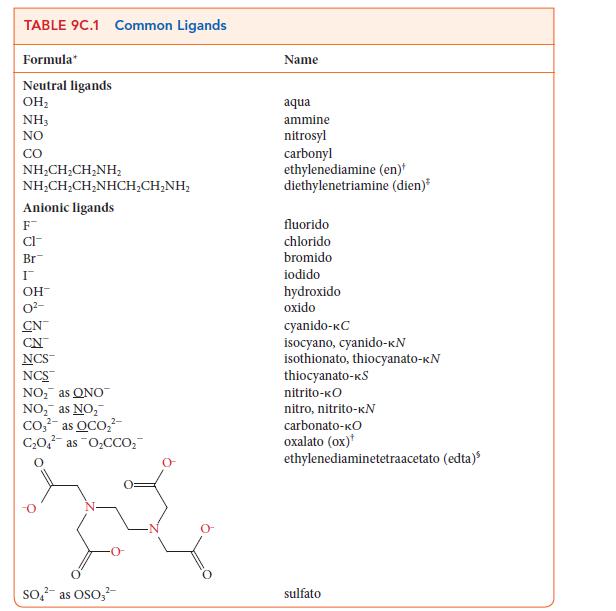
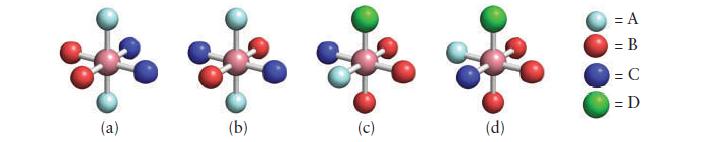
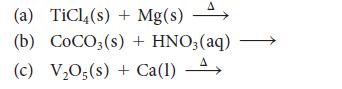

![Examples: [Fe (NCS) (OH),]+ [Fe (NCS) (OH),]+ B. Naming the complex 1. Name the ligands first, and then the](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1704/1/0/1/853659287ddc8fe91704101852651.jpg)