![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


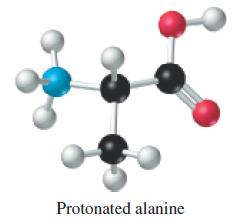
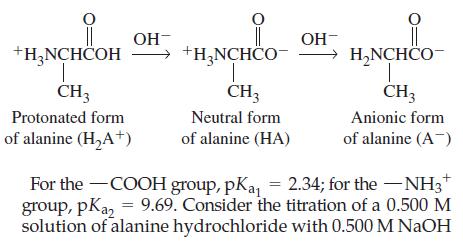
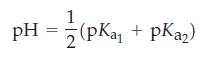
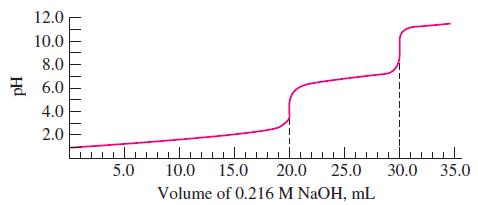
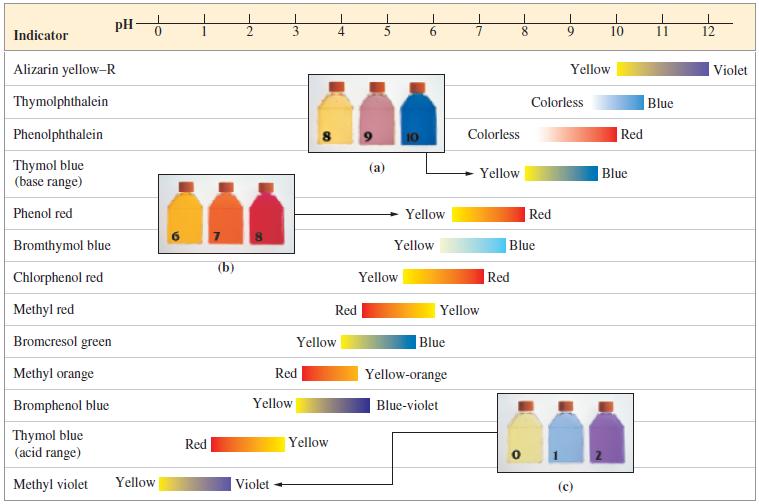


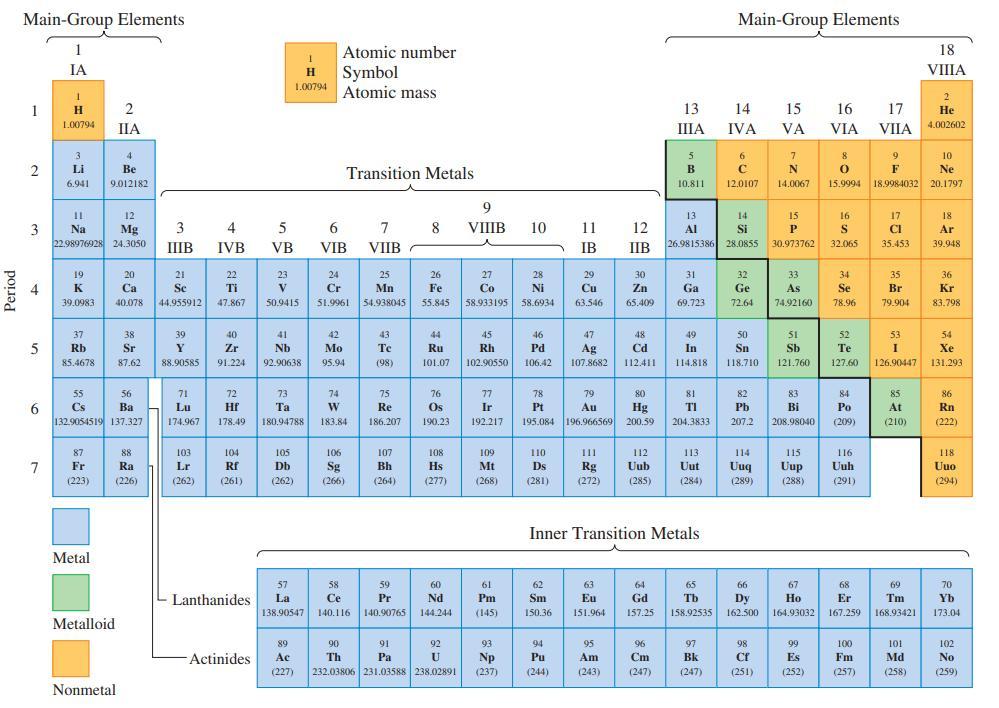
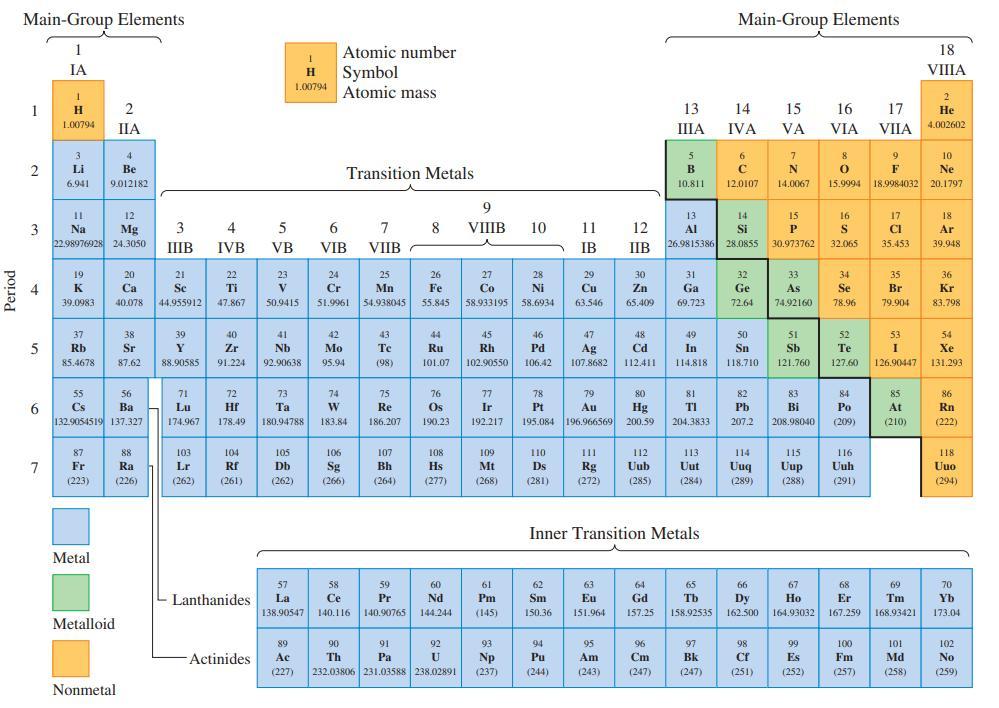
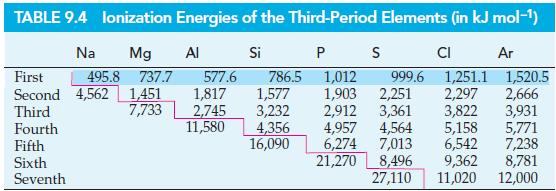
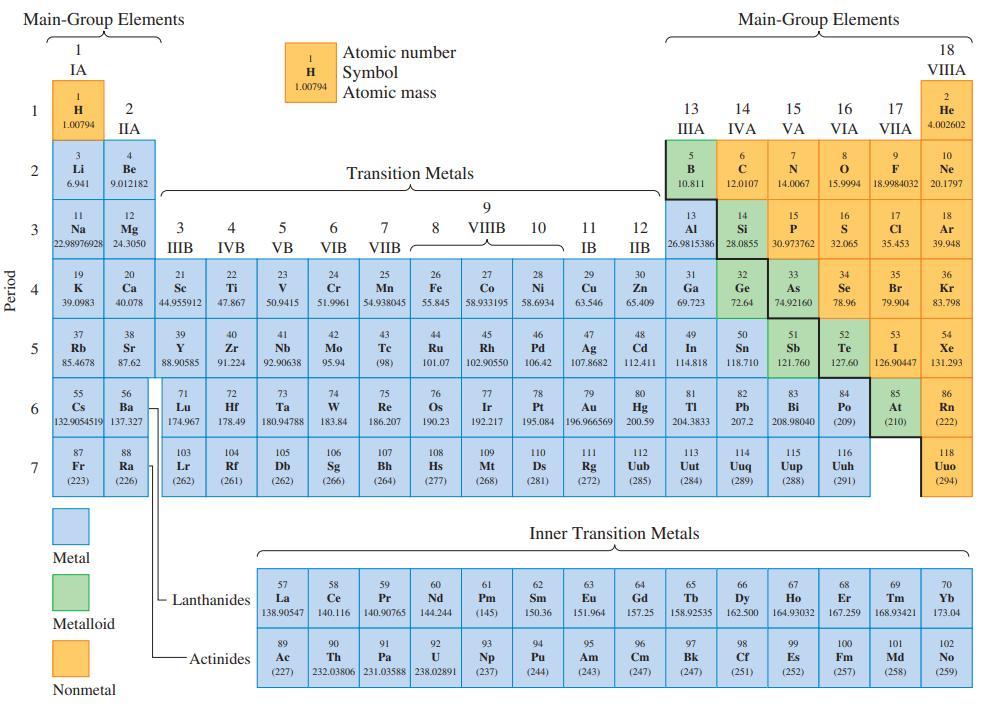
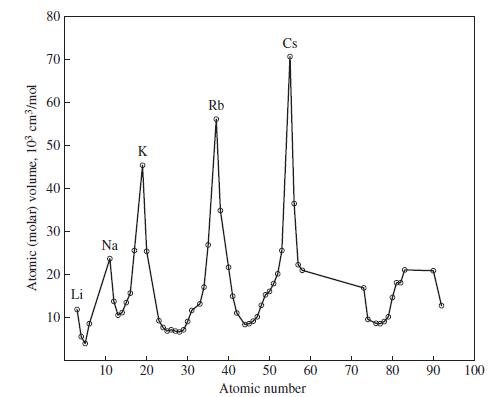
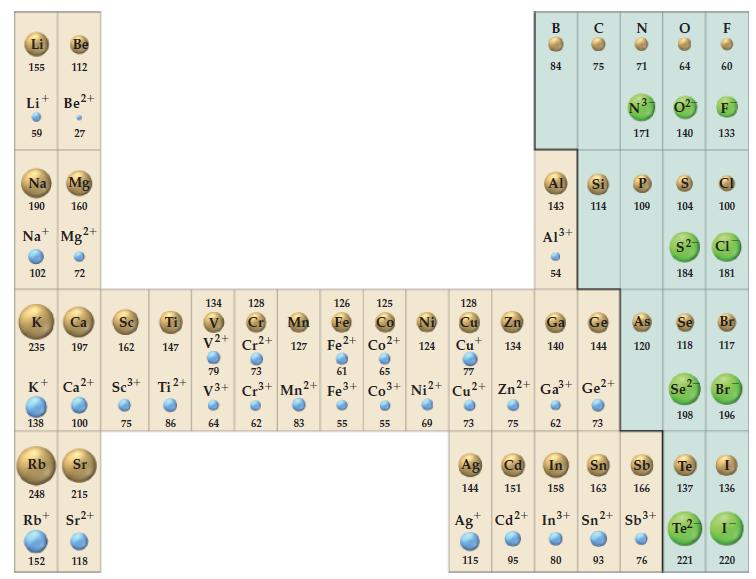
![(a) Tl (b) Z = 70 (c) Ni (d) [Ar]4s (e) a metalloid (f) a nonmetal 1. an alkaline earth metal 2. element in](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/7/8/2/7886550a0848cbe71699782787338.jpg)
