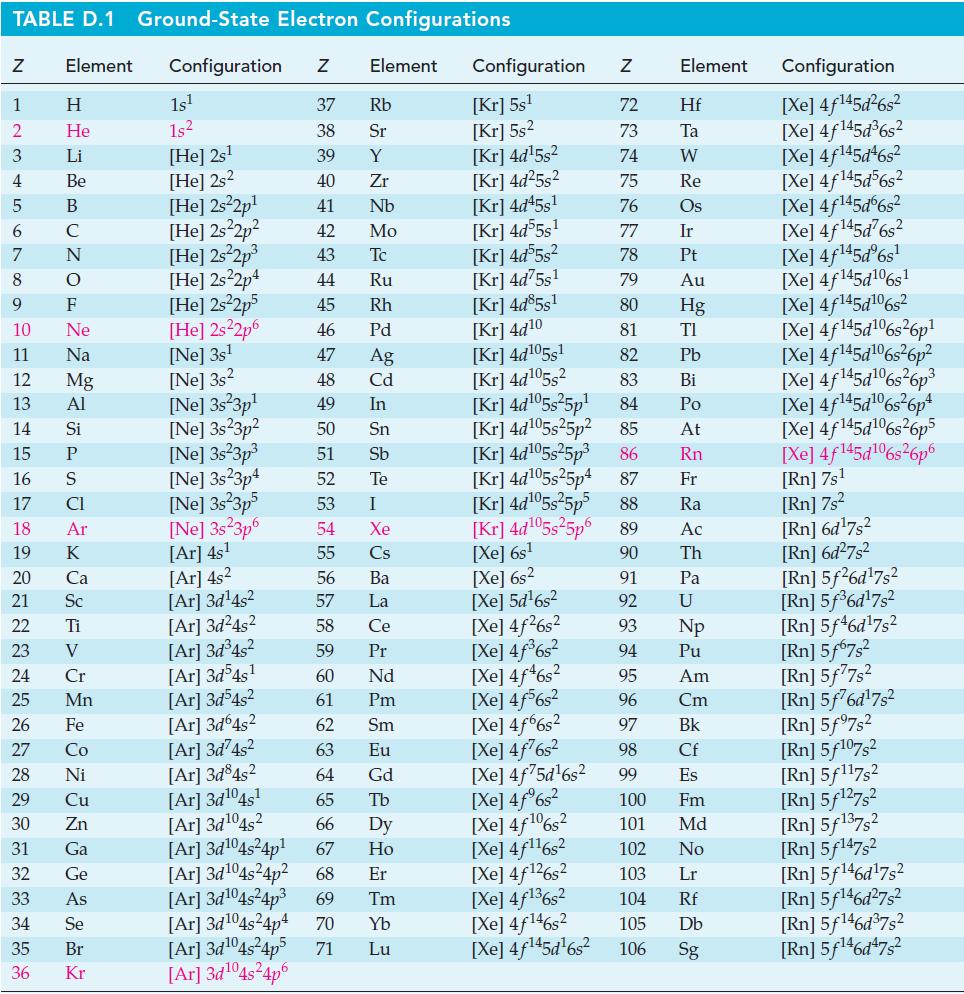
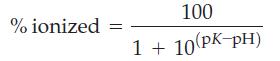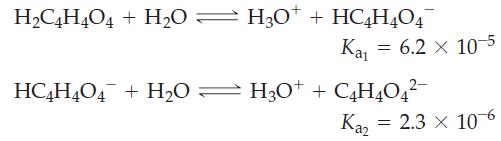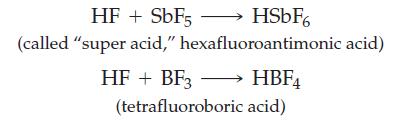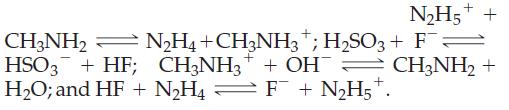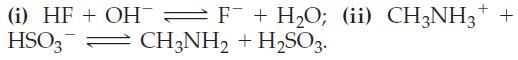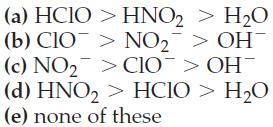![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


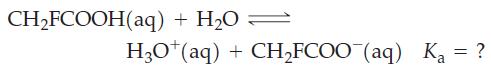
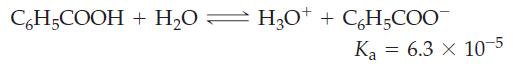
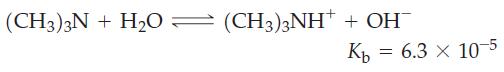
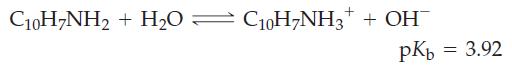
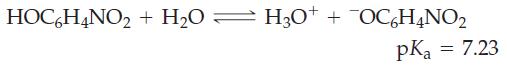
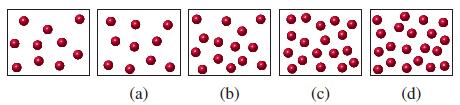
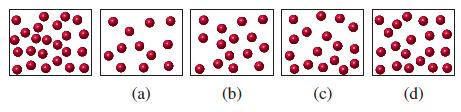
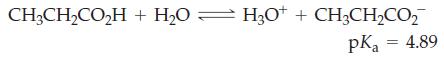
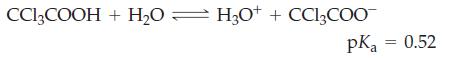
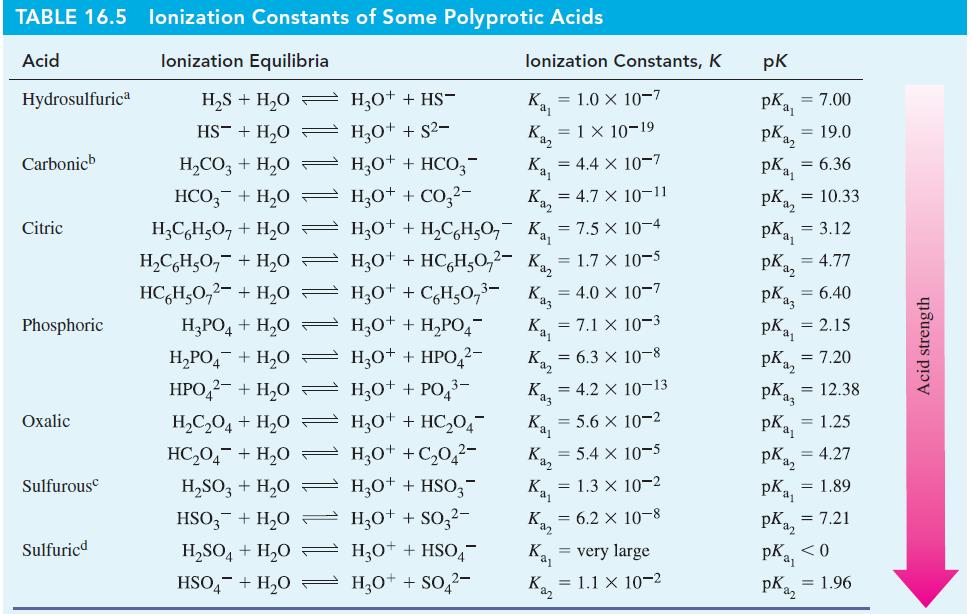
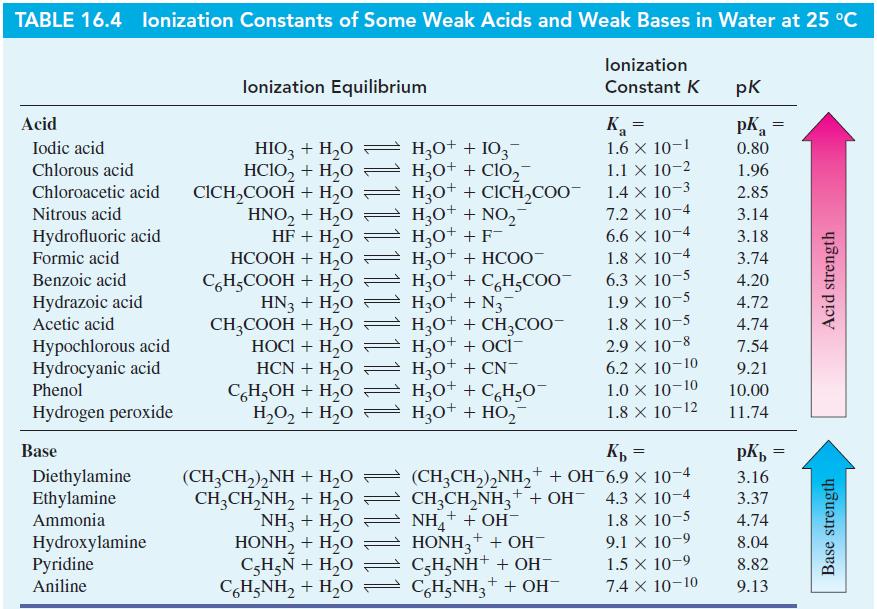
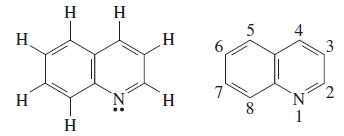

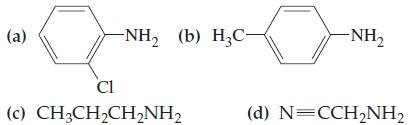
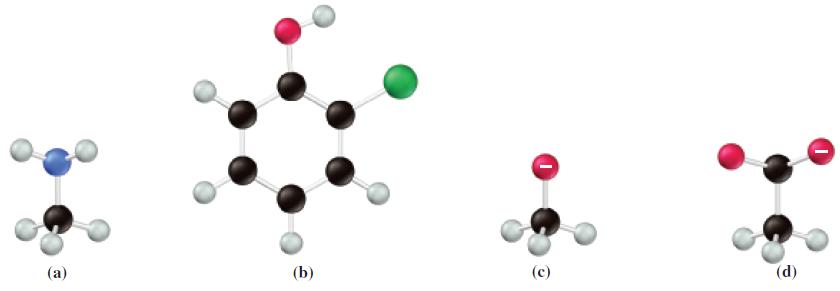
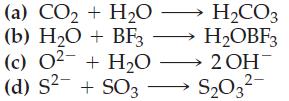
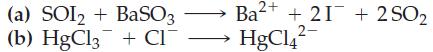
![(a) SO3 + HO HSO4 (b) Zn(OH)(s) + 2OH(aq) [Zn(OH)4] (aq)](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/5/6/2/405655c85e5e4c061..png)
![(a) B(OH)3 + OH (b) NH4 + H3O+ (c) (CH5)2O + BF3 [B(OH)4] NH5+ + HO (C2H5)2OBF3](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/5/6/2/446655c860ee82bc1700562444722.jpg)