![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


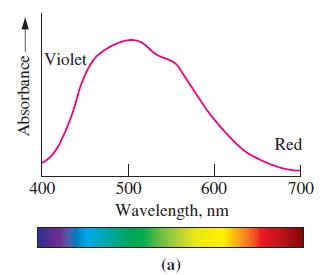
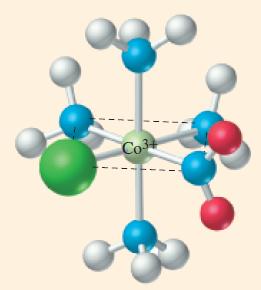
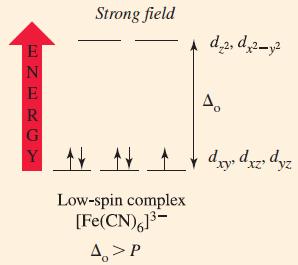
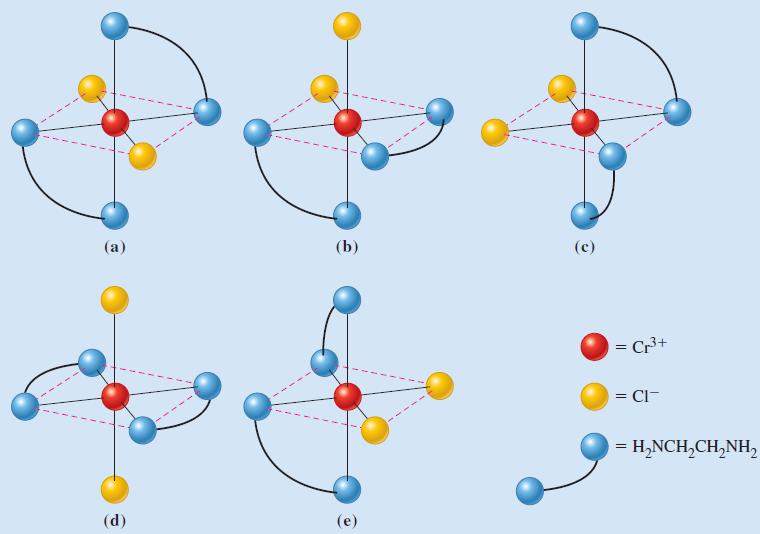
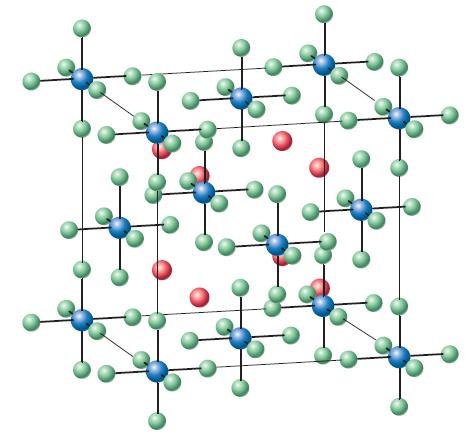
![TABLE 24.5 Some Coordination Compounds Isomer of Cr+ and Their Colors [Cr(HO)6]Cl3 [CrCl(H,O)5]Cl2](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/3/5/1/5646568908c7adb21701351563183.jpg)
![Acidic solution ([H+] = 1 M): +7 +6 MnO (purple) 0.56 V 0.56 V MnO4 (purple) -MnO42- (green) 1.70 V Basic](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/3/3/9/9786568634ae063e1701339977505.jpg)
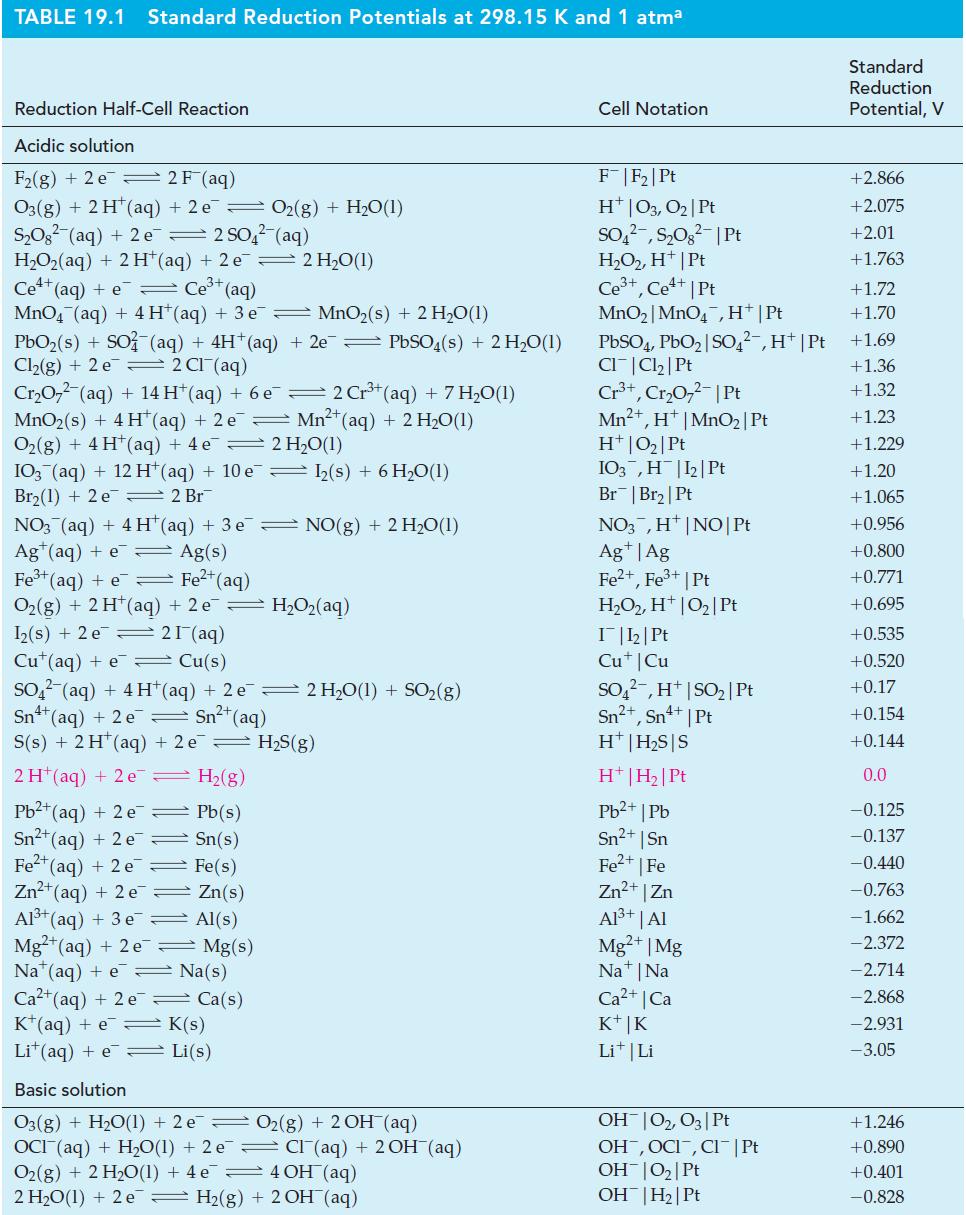
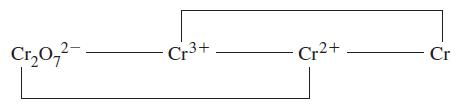
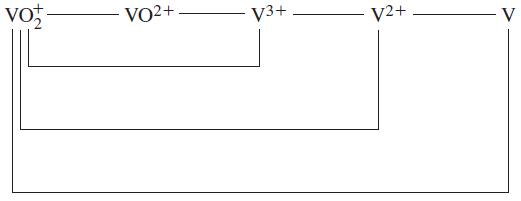
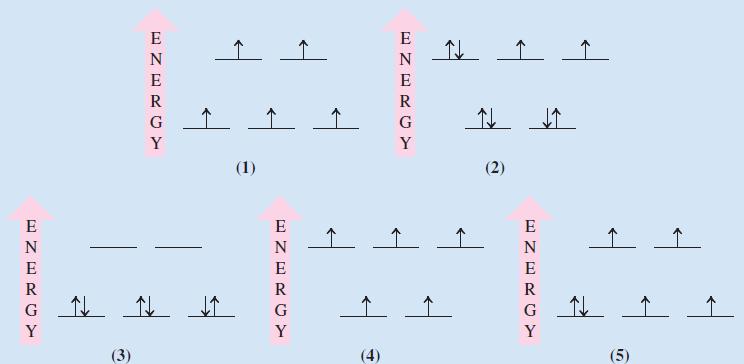
![Kc = [Cr0] [CrO4-][H+] = 3.2 x 1014 (23.19)](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/3/4/0/871656866c7b8f581701340870593.jpg)
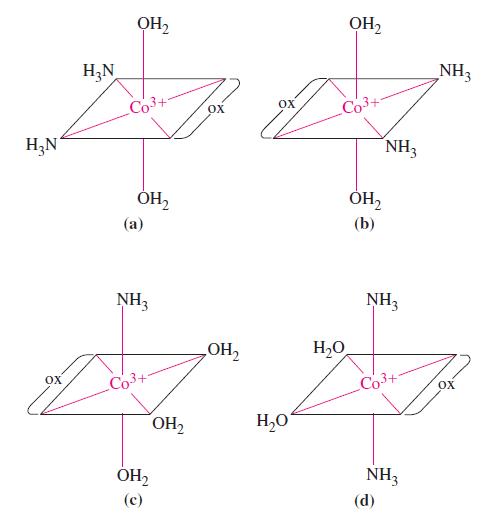
![[Cu(HO) 12+; [Cu(HO)(NH3)3]+; [Cu(en)3]+; [Cu(en)(HO)]2+; [Cu(en)(NH3)4]+.](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/4/1/8/258656995124919d2.png)
![[Cu(HO)4]+ + 4Cl = (blue) [CuCl4] (yellow) + 4HO Kf = 4.2 x 105](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/4/2/5/8046569b28c75db31701425804232.jpg)
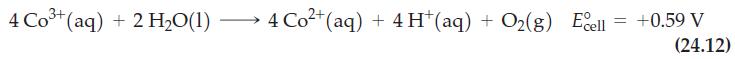
![Ag Ag (0.10 M[Ag(CN)2], 0.10 M CN) ||Ag+ (0.10 M)| Ag](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/4/2/6/5016569b545c9a7e1701426501541.jpg)
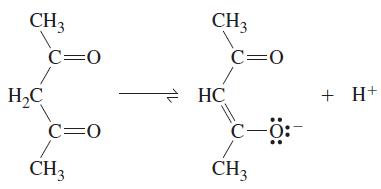
![[Co(HO)6]+ + e [Co(HO)6]2+ + 3 en [Co(HO)6]3+ + 3 en [Co(H,O)%]2+ Calculate E for the reaction [Co(en)3]+ + e](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/4/2/7/7896569ba4d184a01701427788781.jpg)