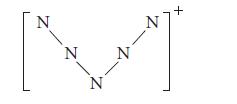
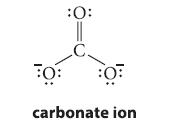
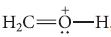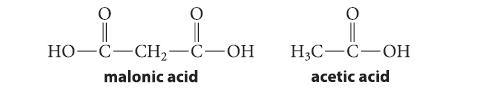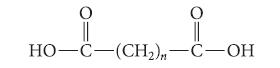![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


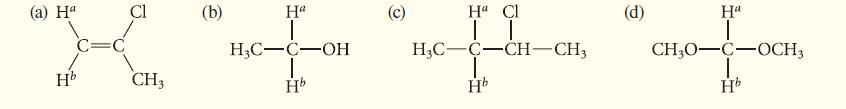
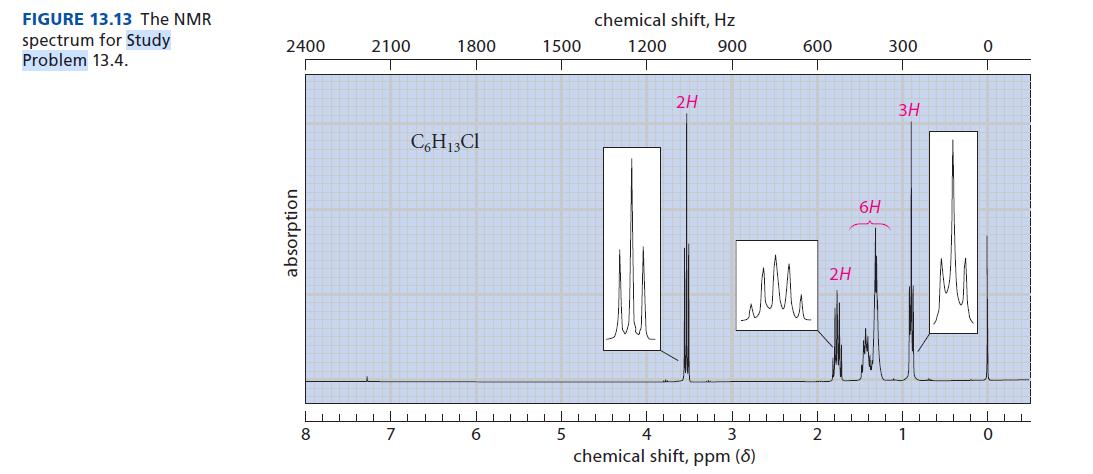
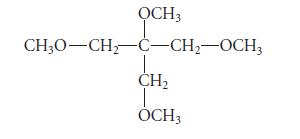
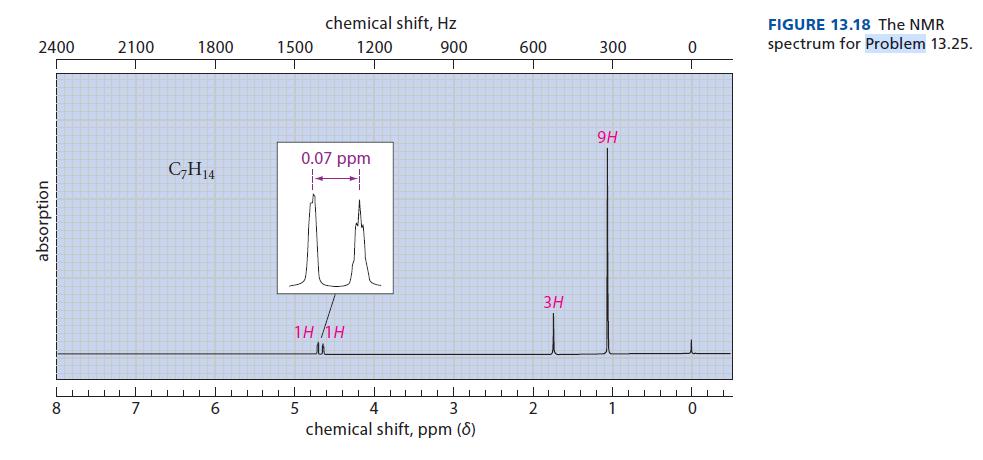
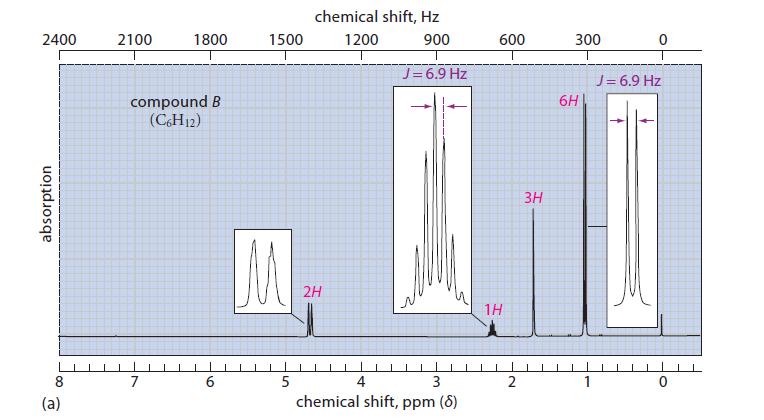
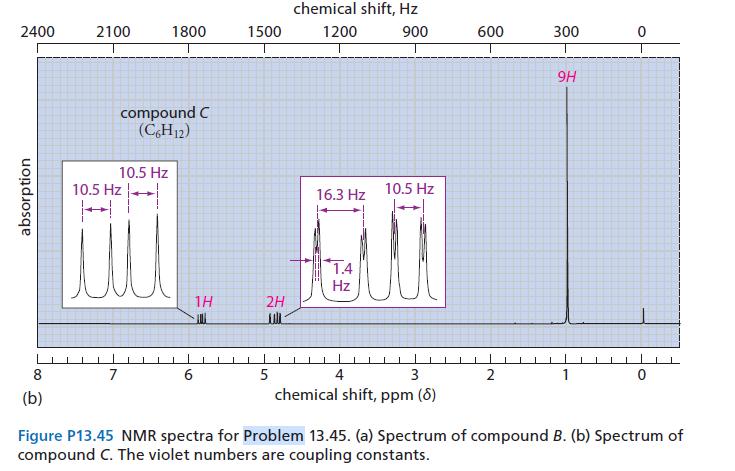

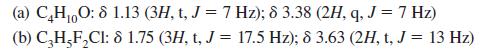
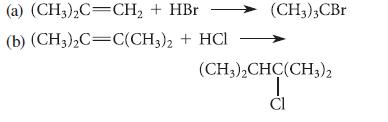
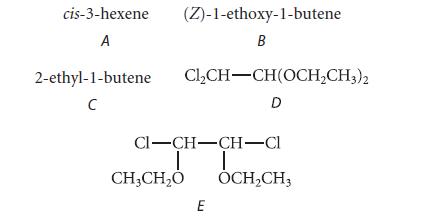
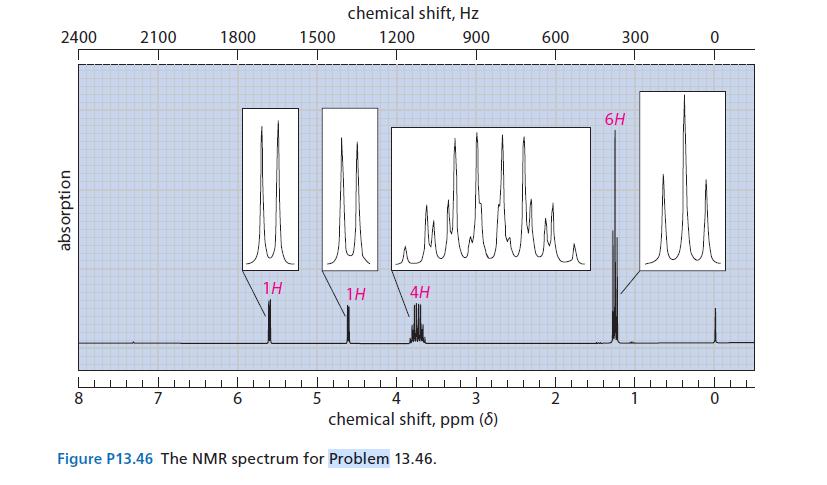
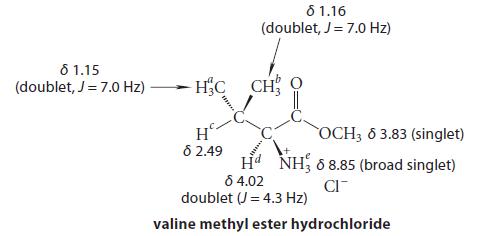
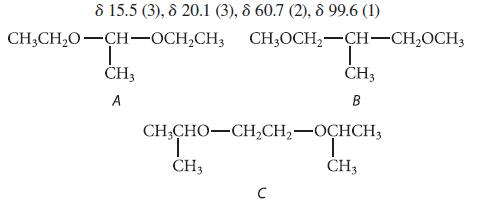
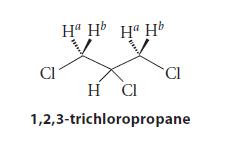
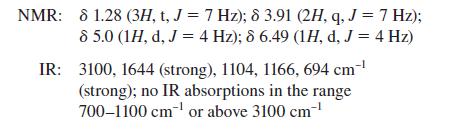
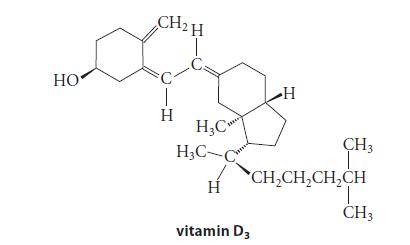
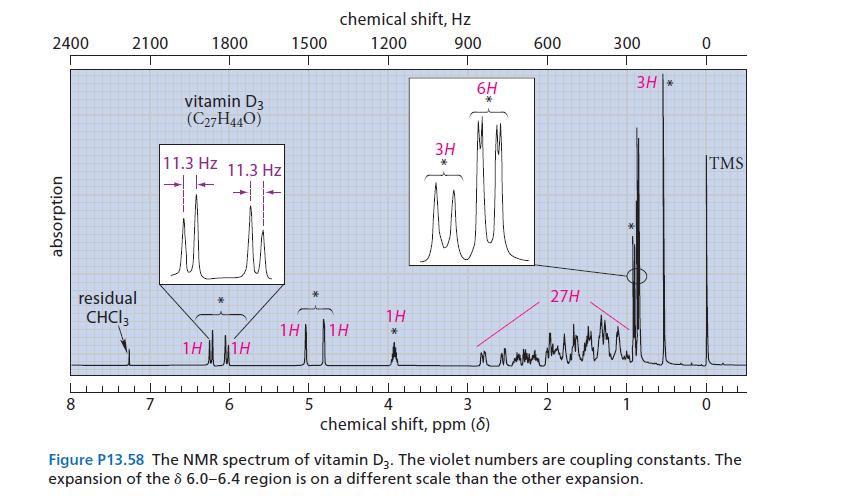
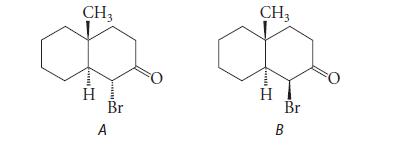
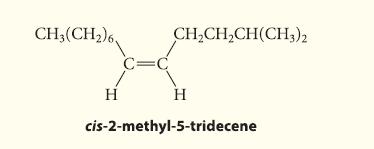
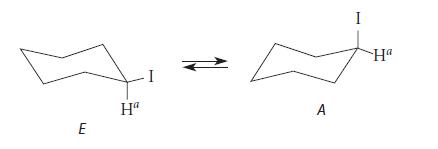
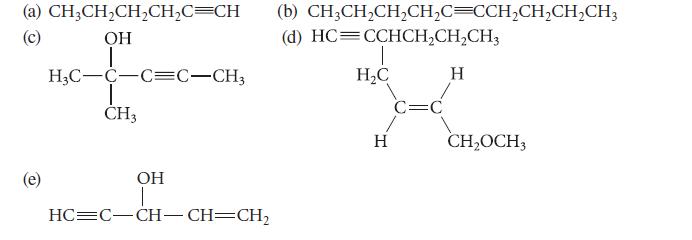
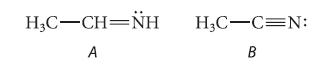
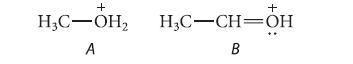
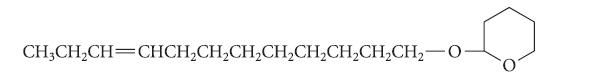
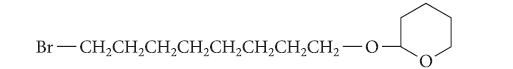
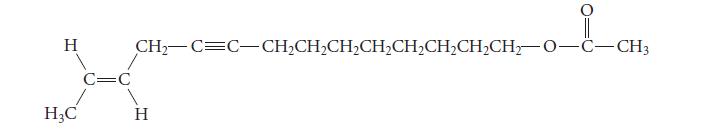
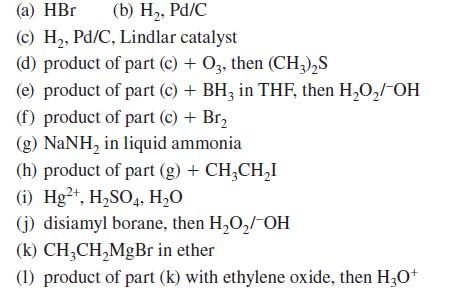
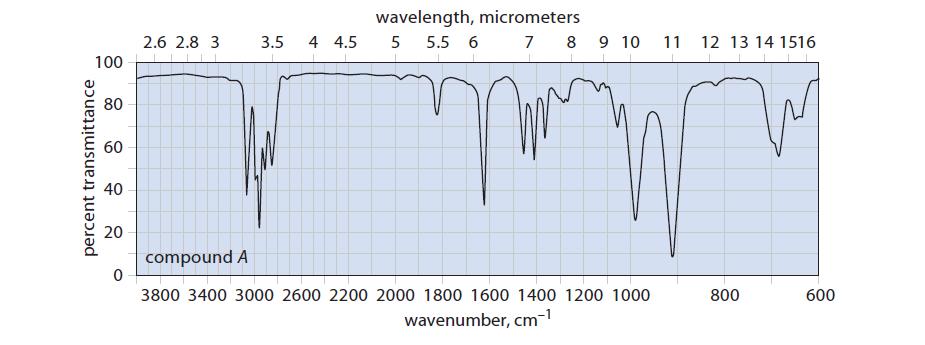
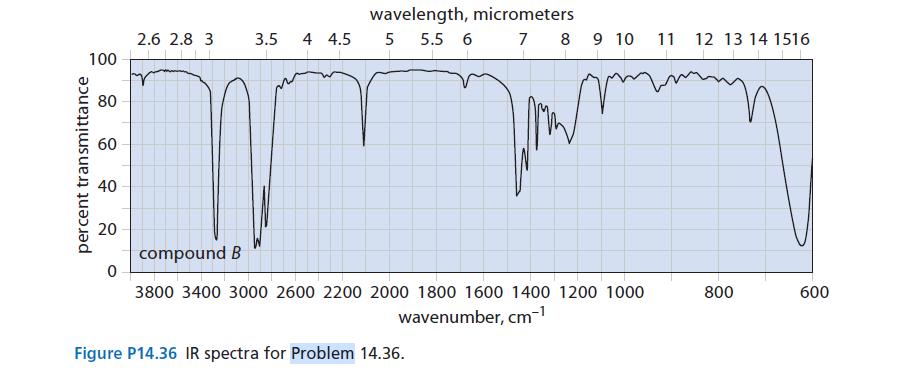
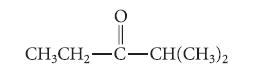
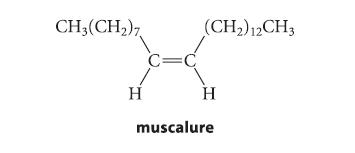
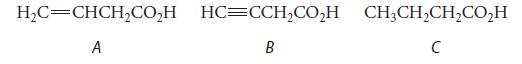
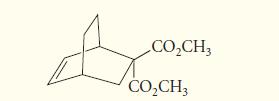
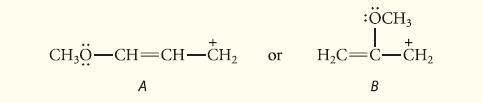

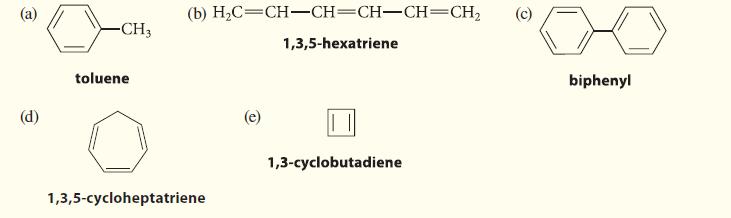
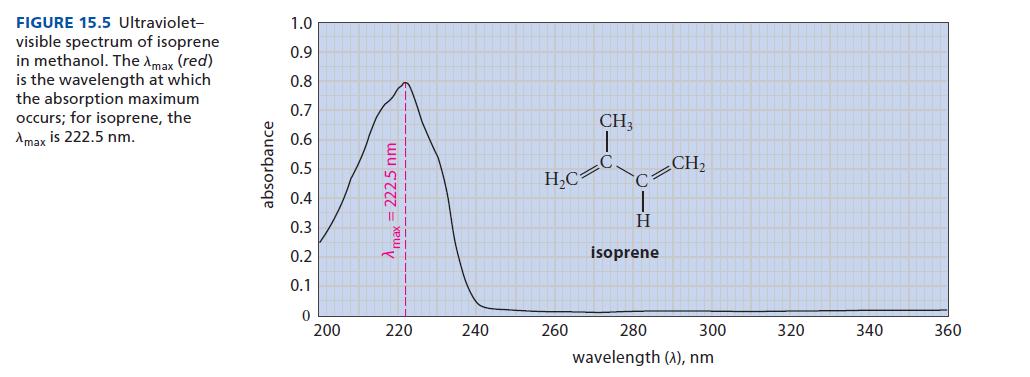
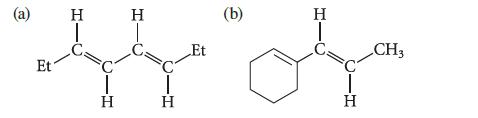
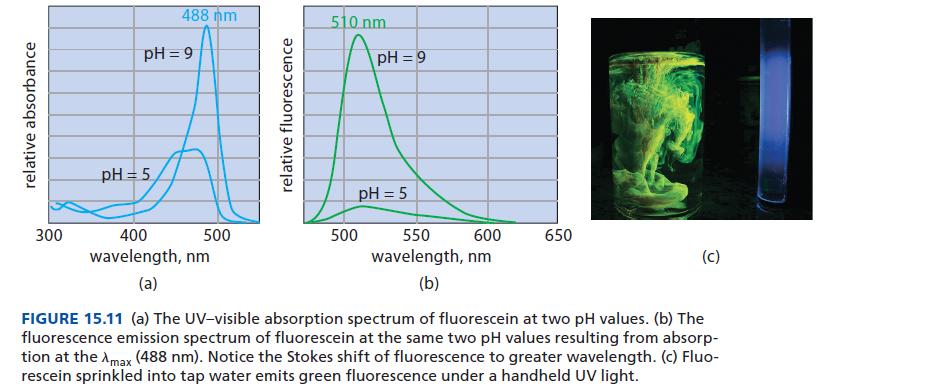
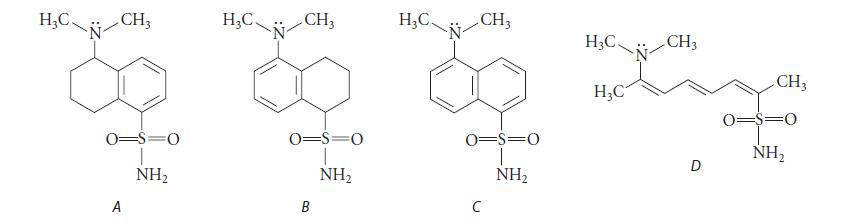
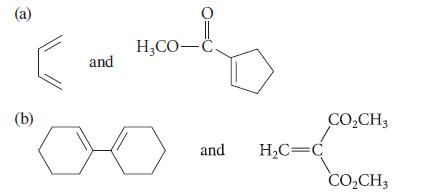
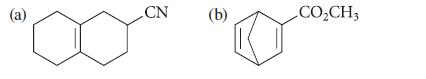
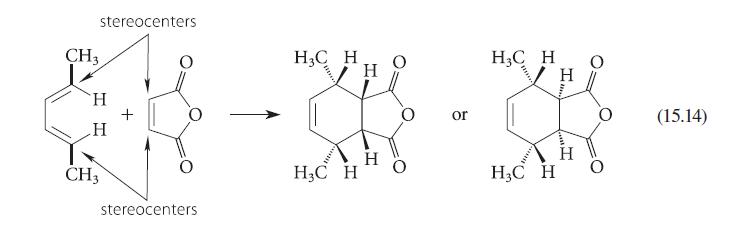
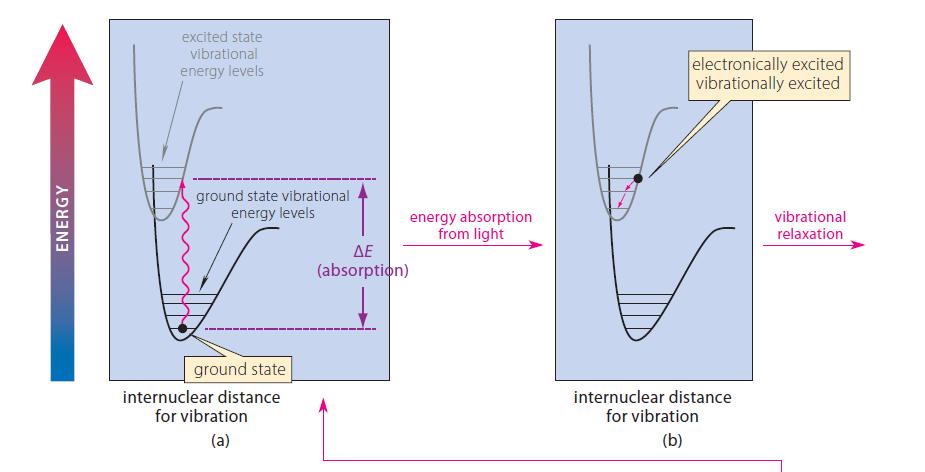
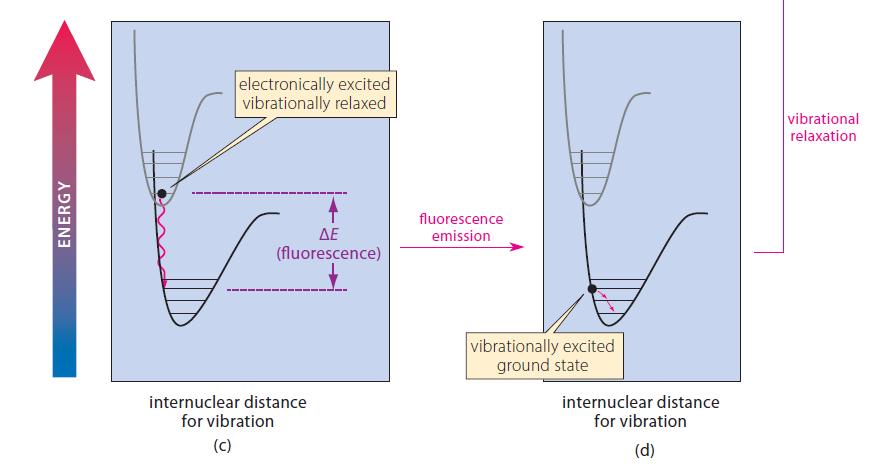
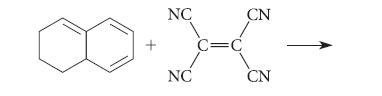
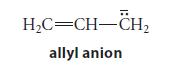
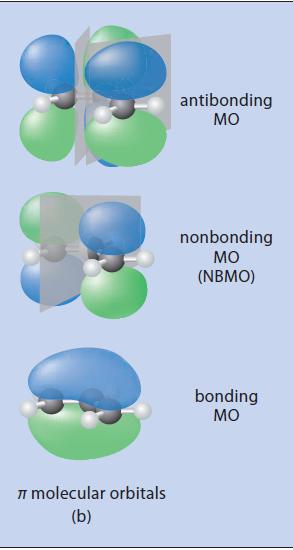
![@ [:c= < :c=] [H_cc=NH carbon monoxide (CH,),CH=CH, (c)[(CH)C=CH-CH H3)2 N: CH3 + CH3 HC-C=NH]](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/4/2/9/3986569c0968fb0b1701429394807.jpg)
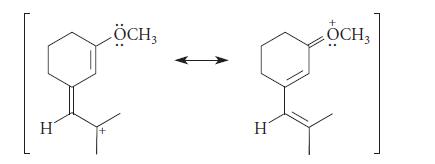
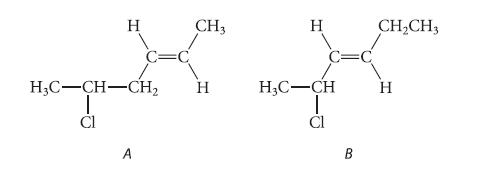
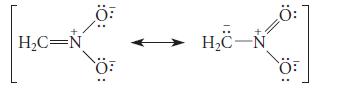

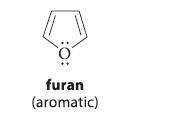
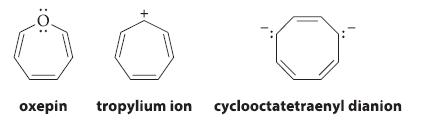
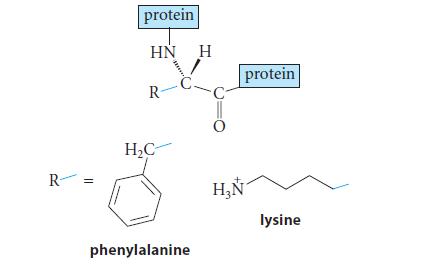
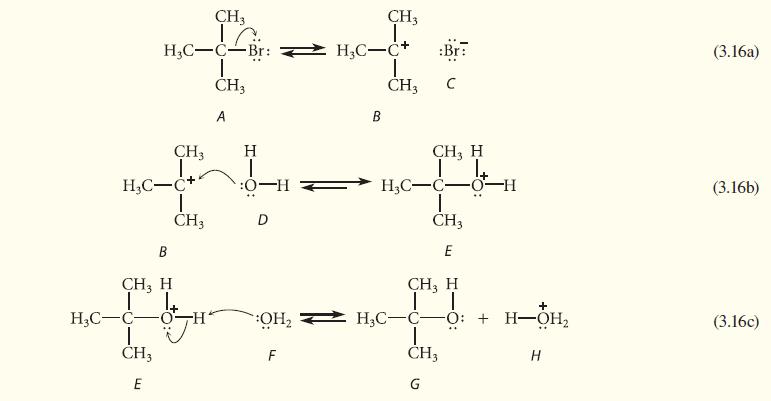

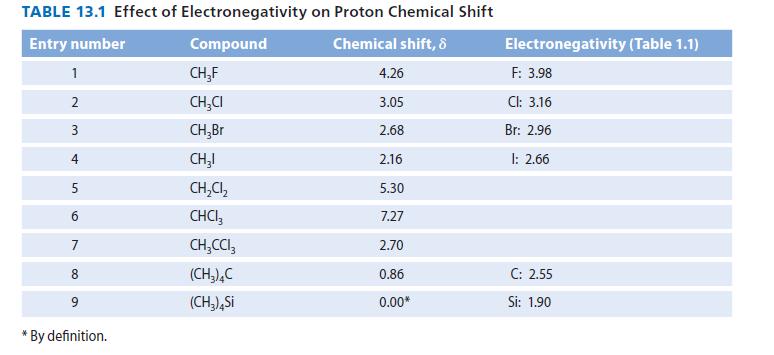
![JAH = [AH] [A] + [AH] [AH] (K[AH]/[H3O+]) + [AH] = [H3O+] K + [H3O+] a 10-PH 10-PK + 10-PH (3.30d)](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/3/1/8/17165680e1b9bff21701318171525.jpg)