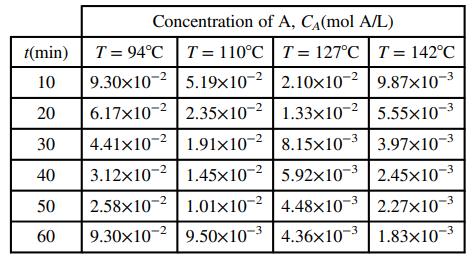
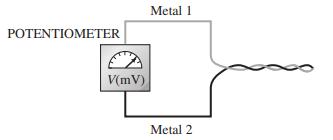
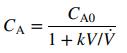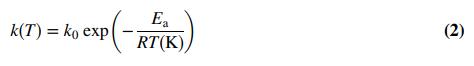Chemistry A Molecular Approach 4th edition Nivaldo J. Tro - Solutions
Unlock the full potential of your chemistry studies with our comprehensive resources for "Chemistry: A Molecular Approach, 4th Edition" by Nivaldo J. Tro. Discover a wealth of knowledge with our online answers key and solutions manual, offering step-by-step answers to enhance your understanding. Access solved problems and in-depth chapter solutions that align perfectly with your textbook. Whether you're preparing for exams with our test bank or seeking detailed instructor manual guidance, our solutions PDF provides everything you need. Best of all, these valuable resources are available for free download, ensuring you have the support to excel in your chemistry journey.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


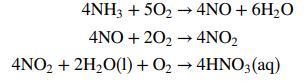
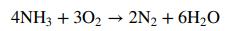
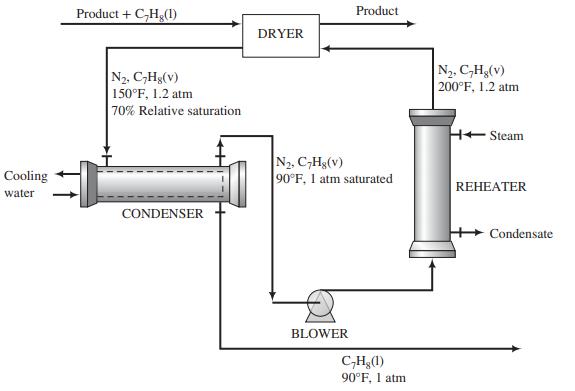
![p*(Tt)[1-fp*(Ta0o)/Po] (1-f)p*(Tao)/Po %3D](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1589/9/8/5/9685ec542b0c03881589985968154.jpg)
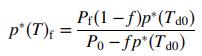

![m = 0.48508 + 1.55171@ – 0.1561@? (5.3-11) T; = T/T. (5.3-12) m(1 – VT.)]* a = |1 + (5.3-13)](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1589/9/7/3/8605ec513649dc451589973858626.jpg)



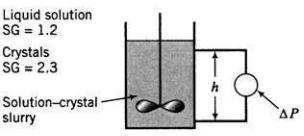
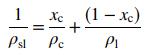
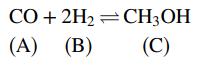
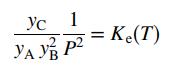
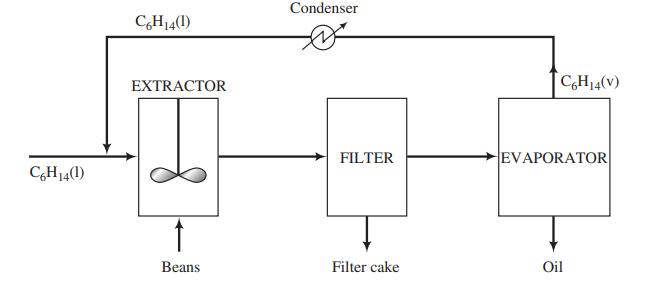
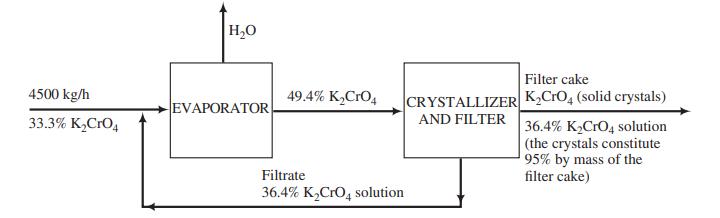
![m,(kg Cr/h) [no water] 95% of Cr in feed to unit Wastewater m(kg/h) 0.0515 kg Cr/kg m2(kg/h) < 4500 kg/h m;(kg/h) m(kg/h) m,(kg/h) To waste lagoon](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1589/9/0/1/4145ec3f866e48461589901411043.jpg)